 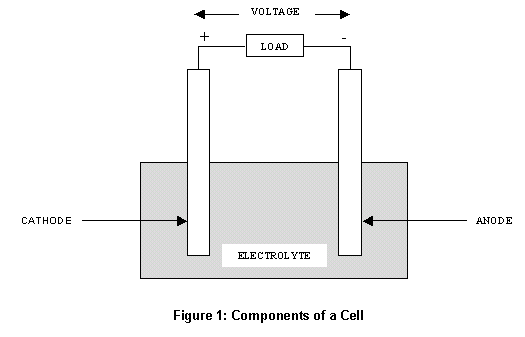
Hence, zinc oxidises in Zn² + become the agar gel milky white colour instead of bluish colour. In above reaction, the zinc has turned the anode as it is a more sensitive metal than iron. The figure 4 shows the half zinc plated iron nail The blue area illustrates the position of the anode when the reddish-pink illustrated the position of the cathode. Hence, the indicators can cause to occur the position of the anodes and cathodes in the galvanic corrosion.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
